UK Volunteers Begin Vaccination Against H5N1 Bird Flu
The first volunteers in the United Kingdom have been immunised with a vaccine designed to protect against a potential bird flu pandemic.
This vaccine specifically targets the H5N1 influenza strain, which has caused severe outbreaks in bird populations globally and has also spread to some mammalian species.
The current risk to humans remains low, according to the UK Health Protection Agency, as nearly all human cases have been linked to close contact with infected animals.
The vaccine employs the same mRNA technology used in existing Covid-19 vaccines, a method that scientists say allows for rapid and large-scale production in the event of a pandemic.
The clinical trial aims to recruit individuals who work in the poultry industry or are aged over 65, as these groups are considered most at risk.
Clare Howard, a resident of Hampshire who has kept chickens for many years, was among the first volunteers to receive the vaccine in this new H5N1 trial at a clinic in Southampton.
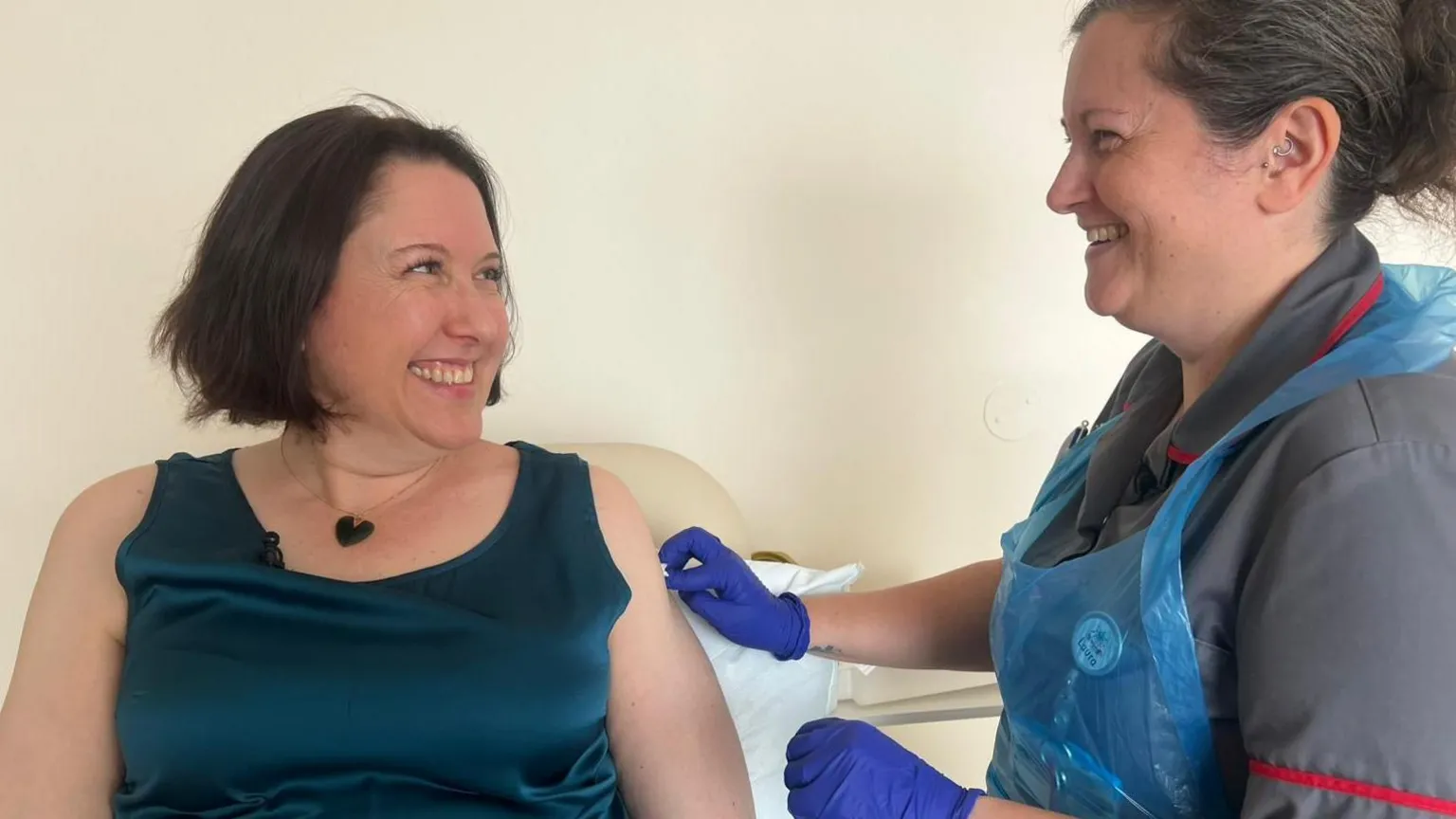
"It was quite easy and it could be something that ultimately proves incredibly important," she said.
The extensive trial plans to involve 4,000 volunteers, with three-quarters recruited across 26 sites in England and Scotland, and the remainder in the United States.
Trial Leadership and Virus Evolution
Dr Rebecca Clark, the trial's national coordinating investigator based at Layton Medical Centre in Blackpool, noted that the H5N1 strain is "evolving and spreading across animal species."
"Although it does not yet move easily between humans, we have to treat human-to-human transmission as a real possibility," she said.
"This trial is our proactive attempt to shield against that possibility, and any future pandemic that could emerge from it."
Since 2024, there have been 116 confirmed human cases worldwide, almost all connected to close contact with infected animals.
The study will assess whether the vaccine is safe and capable of eliciting a strong immune response. If successful, it could be approved for use when necessary.
Professor Lucy Chappell, chief scientific adviser at the Department of Health and Social Care and chief executive officer of the National Institute for Health and Care Research, stated that the trial is "bolstering our pandemic resilience."
Manufacturing Capacity and Technology Advantages
If required, the vaccine would be produced at Moderna's new manufacturing facility in Harwell, Oxfordshire, which currently produces Covid-19 vaccines for the UK.
This plant has an annual production capacity of 100 million doses, which could be increased to 250 million doses during a pandemic.
Advantages of mRNA Vaccines Over Traditional Methods
Traditional flu vaccine production involves growing the virus in eggs, a process that can be problematic when dealing with virulent avian flu strains that can kill the eggs used during manufacturing.
During the Covid-19 pandemic, mRNA vaccines demonstrated high effectiveness in preventing severe illness and could be produced and modified rapidly as viral strains evolved.
Flu pandemics are inevitable, although the timing of the next global outbreak remains uncertain. The influenza virus constantly evolves, necessitating a new flu vaccine annually.
A flu pandemic occurs when the virus undergoes a significant shift, rather than a gradual drift, resulting in a strain to which humans have no natural immunity.
The last pandemic in 2009, known as swine flu, was relatively mild. In contrast, the Spanish flu pandemic following the First World War resulted in approximately 50 million deaths worldwide.
It is currently unknown whether H5N1 will be the strain responsible for the next flu pandemic.
Previous Vaccine Efforts and Case Statistics
There have been other experimental vaccines targeting H5N1. One such trial took place in Oxford in 2006; although the vaccine was safe, it did not demonstrate strong effectiveness.
Since 2003, approximately 1,000 confirmed human cases of H5N1 have been reported to the World Health Organization, with nearly half resulting in fatalities.
More recently, a strain circulating in the United States caused milder symptoms, primarily eye inflammation.
Funding and Global Access
In August 2025, the US government reduced funding for mRNA vaccines by $500 million after Health Secretary Robert F. Kennedy Jr., a vaccine sceptic, stated that "mRNA technology poses more risks than benefits" for respiratory viruses.
The Coalition for Epidemic Preparedness Innovations (CEPI) has provided £40 million in funding for the current trial.
As part of the agreement, Moderna has committed to ensuring rapid and affordable vaccine supply to low- and middle-income countries in the event of a future pandemic.
CEPI indicated that this commitment aims to prevent the "vaccine nationalism" observed during the Covid-19 pandemic, when millions remained unprotected as wealthy countries secured early vaccine supplies.






