Breakthrough Alzheimer's Drugs Questioned
An influential analysis has concluded that so-called "breakthrough" Alzheimer's drugs are unlikely to provide meaningful benefits to patients. Researchers stated that the impact of these drugs is "well below" what is necessary to significantly improve the lives of those with dementia.
However, this report has sparked a strong backlash from other respected scientists who argue that the analysis is fundamentally flawed.
Currently, the NHS does not fund these drugs, and an 18-month treatment course costs approximately £90,000 if paid for privately. This price places the drugs out of reach for most people, raising the question of whether they are worth the expense even for those who can afford them.
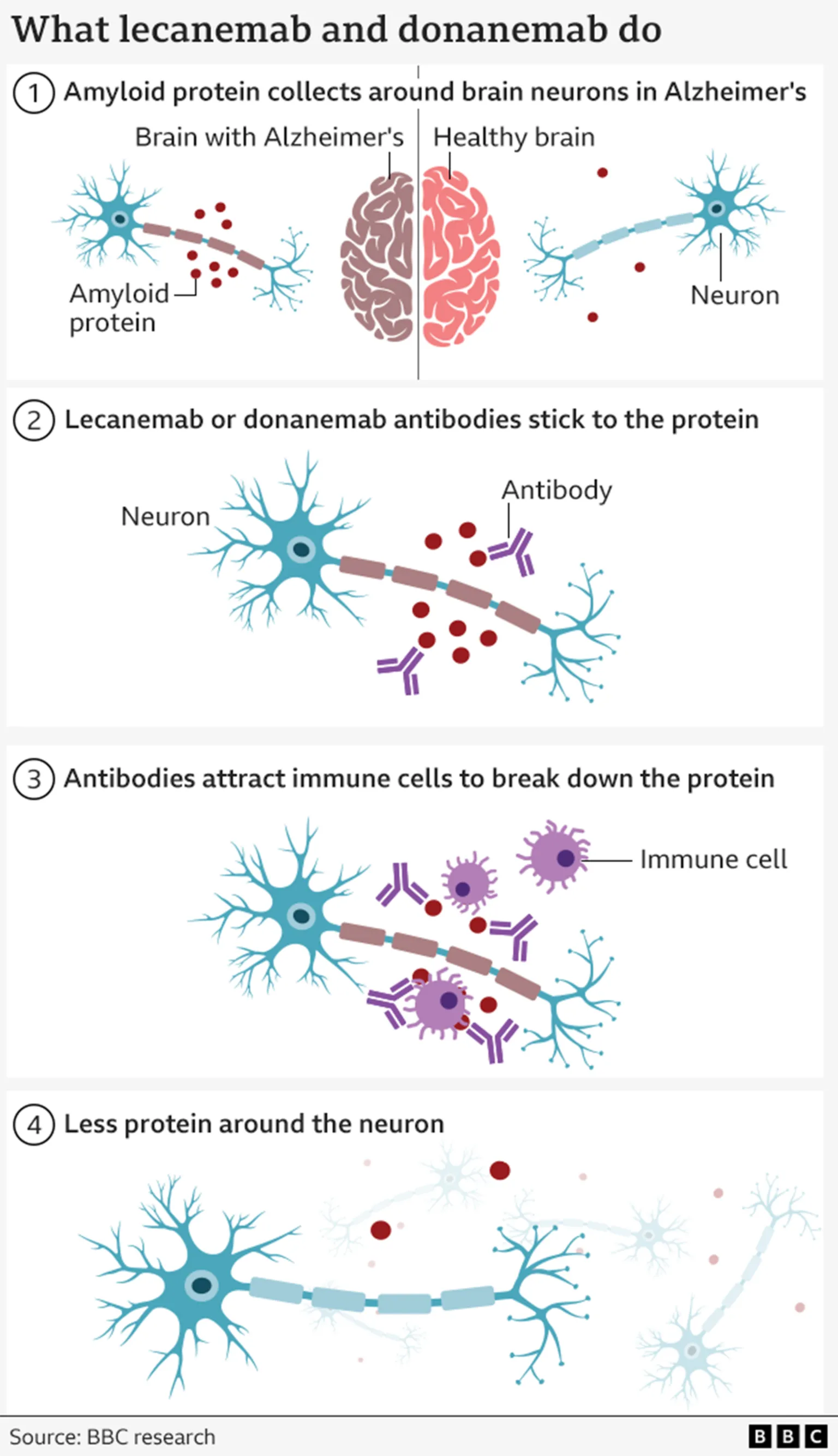
The drugs target beta amyloid, a sticky protein that accumulates between brain cells in Alzheimer's disease.
Falling Short of Expectations
These treatments use engineered antibodies, similar to those the immune system produces to combat viruses or bacteria, designed to identify and clear amyloid from the brain.
For many years, this approach failed to yield results. However, recent trials of two drugs, donanemab and lecanemab, demonstrated that they could slow cognitive decline, marking the first time any medication has slowed brain degeneration in Alzheimer's disease.
The Cochrane Collaboration, known for its rigorous and independent medical data analyses, reviewed 17 studies involving 20,342 volunteers that examined drugs aimed at removing amyloid from the brain.
Overall, the collaboration concluded that while the approach does slow Alzheimer's disease progression, the effect is insufficient to produce a meaningful difference for patients.
Additionally, these medicines carry risks such as brain swelling and bleeding. They require administration every two to four weeks and come at a high financial cost.
One of the report's authors, Professor Edo Richard, a neurology professor at Radboud University Medical Centre in the Netherlands, treats dementia patients in his clinic.
When asked what he would tell his patients, he responded:
"I would tell them, I think you will probably not benefit from these drugs and they're burdensome for you and your family. I think it's extremely important that we're honest to our patients about what they can expect, I'm always wary to avoid giving people false hope."
He emphasized the need to explore alternative treatment methods for Alzheimer's disease, such as targeting brain inflammation.
Support and Criticism
The report's findings have been supported by long-standing critics of these drugs.
Professor Robert Howard of University College London (UCL) described the hype surrounding these drugs as "unfortunate and unfair" to families affected by dementia, stating that it is "not supported by robust science and that will have raised false hopes."
However, the methodology of the analysis has generated heated debate.
The research team argues that since all the drugs reviewed remove amyloid from the brain, their analysis effectively assesses whether this approach works.
Conversely, other experts contend that differences in how each drug functions are significant, and it is inappropriate to group older experimental drugs with newer ones that have demonstrated efficacy.
Professor Bart De Strooper from the UK Dementia Research Institute at UCL commented:
"The review does not clarify the evidence, it blurs it. The flaw in this review is fundamental. Many early programmes failed, but newer antibodies have delivered modest yet real clinical benefit."
Dr. Richard Oakley of the Alzheimer's Society added:
"It's essential that we interpret this review with nuance and avoid taking a sledgehammer to decades of pioneering scientific study."
Access and Funding Challenges
Currently, the only way to obtain these drugs in the UK is through private payment, making them inaccessible to most people.
The National Institute of Health and Care Excellence (NICE), responsible for determining which drugs the NHS will fund, has previously rejected these treatments. However, it is currently reviewing the evidence again, taking into account the burden placed on unpaid carers.






